Summary :
EMB agar is a commonly used culture media in microbiology laboratories. It is a useful tool for identifying and isolating enteric pathogens, as well as for screening water samples for fecal contamination. In this article, we explore its principle, its preparation, its interpretation and its different uses.
Ⅰ. Overview
Eosin Methylene Blue Agar, EMB agar, is a selective and differential culture media used for the isolation of gram-negative bacilli in foods, dairy products and clinical samples.
This agar provides both selective and differential properties, making it a valuable medium in microbiology laboratories.
- As a selective medium, EMB agar contains inhibitors, such as eosin and methylene blue, that suppress the growth of gram-positive bacteria.
- As a differential medium, it can differentiates between different types of gram-negative bacteria based on their ability to ferment lactose.
It was originally formulated in 1916 by Holt-Harris and Teague, then the agar was modified by Levine in 1918 which removed the sucrose from the formula and increased the lactose content. Today, EMB Agar is a combination of both formulas.
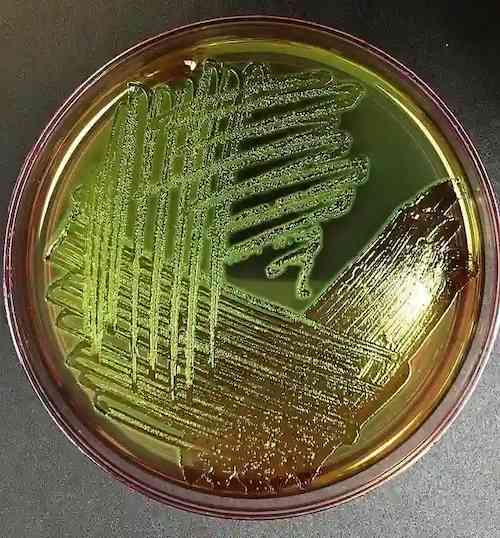
E.coli in EMB Agar
Ⅱ. Preparation and composition
Suspend the components, dehydrated powder of EMB Agar, in water (36 grams in 1000 ml of purified/distilled water).
The medium is boiled for a few seconds until the ingredients are completely dissolved. Sterilize in an autoclave at 15 lbs (121°C) pressure for 15 minutes. avoid overheating.
Cool to 45-50°C and stir the medium to oxidize the methylene blue (i.e. restore its blue color) and suspending the flocculant precipitate. Pour into Petri dishes.
Composition of EMB Agar |
|||
|---|---|---|---|
| Ingredients | Gram/liter | ||
| Animal Tissue Peptic Digest | 10,000 | ||
| Dipotassium Phosphate | 2,000 | ||
| Lactose | 5,000 | ||
| Sucrose | 5,000 | ||
| Eosin - Y | 0.400 | ||
| Methylene blue | 0.065 | ||
| Agar | 13.500 | ||
Ⅲ. Principle of EMB Agar
Selectivity : Eosin Methylene Blue Agar contains two dyes, eosin and methylene blue, which inhibit the growth of gram-positive bacteria. These dyes have an inhibitory effect on the growth of gram-positive organisms while allowing the growth of gram-negative bacteria.
Differential : EMB agar differentiates between different types of gram-negative bacteria based on their ability to ferment lactose. Lactose-fermenting bacteria produce acid during lactose fermentation, which leads to a color change on the agar.
- Strong lactose fermenters: E. coli is a strong lactose fermenter and produces colonies with a distinct green metallic sheen on EMB agar.
- Weak lactose fermenters : Some other lactose-fermenting bacteria (slow-fermenting lactose) may produce brown-pink colonies.
- Non-lactose fermenters : such as Shigella and Salmonella, do not produce acid and form clear, colorless or amber colonies that are easily distinguished from coliforms.
The phosphate buffers the medium.
Note: The distinctive metallic sheen produced by E. coli on this culture media is due to acid production resulting in an amide bond between eosin and methylene blue, other coliforms not producing enough acid to cause this reaction.
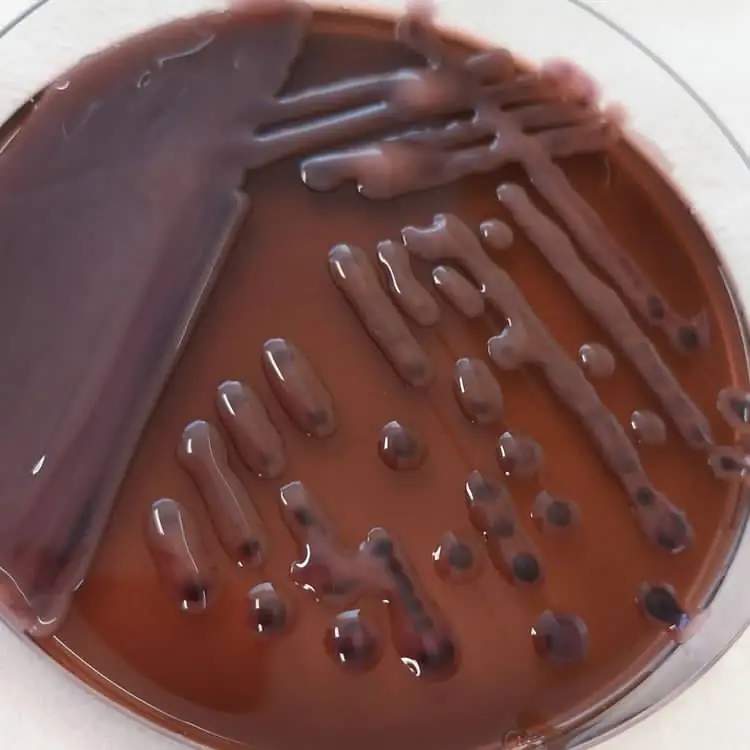
Klebsiella in EMB Agar
Ⅳ. Results and Interpretation
The interpretation of EMB agar involves observing the colony morphology and color changes of bacterial growth on the medium (In general, after 24 hours at 37°C aerobically). Here are the general interpretations:
First, you observe whether there is any growth of colonies on the EMB agar. If there is no growth, it could indicate :
- The absence of bacteria.
- Presence of gram-positive bacteria (which are inhibited by the selective components of the medium)
- The presence of bacteria that are difficult to grow on this medium (bacteria with special needs).
If there is growth on the EMB agar, you then examine the color of the colonies:
- Strong lactose fermenters, like E. coli, typically produce colonies with a intense green metallic sheen.
- The pink color indicates a lesser degree of lactose fermentation compared to strong fermenters.
- Colorless colonies on EMB agar typically indicate non-lactose fermenting bacteria.
Note : Further confirmatory tests or identification methods should be employed to accurately identify specific bacterial species.
| Organisms | Growth |
|---|---|
| E. coli | Blue-black colonies with green metallic sheen |
| Klebsiella | Pink and mucoid colonies |
| Enterobacter aerogenes | Good growth; pink, dull |
| Proteus | Variable in size, blue-green to blue or salmon in color, most stumps being black in the center or over their entire area |
| Salmonella | Colorless Colonies |
| Shigella | Colorless Colonies |
| Pseudomonas | Colorless Colonies |
| Gram positive bacteria | Inhibition (partial to complete) |