Summary :
- Ⅰ. Introduction
- Ⅱ. Parts
- Ⅲ. Principle
- Ⅳ. Fuel Sources
- Ⅴ. Types of Bunsen Burners
- Ⅵ. What are the types of flame on a Bunsen burner ?
◉ Introduction
A Bunsen burner is a type of gas burner that produces a safe, smokeless, hot, non-luminous flame that can be used for various scientific experiments.
The flame is created by the gas and oxygen being mixed in a controlled environment, which allows precise regulation of the size and heat of the burner.
The Bunsen burner consists of a flat base with a vertically extending straight tube, called a barrel or chimney. Natural gas (mainly methane) or a liquefied petroleum gas such as propane or butane is supplied at the bottom of the chimney, it is connected to a gas valve using a rubber tube.
The burner was named after Robert Bunsen, a German scientist who designed it in 1857 with his laboratory assistant, Peter Desaga.

Bunsen burner
◉ The basic function of a Bunsen burner in the laboratory is heating, sterilization and combustion.
◉ Bunsen burner parts
It consists of several parts, each of which plays an important role in the functioning of the burner. The main parts of a Bunsen burner include :
- Base : It is a wide and heavy part of variable shape which provides support for the burner. It is connected to a side tube called the gas tube.
- Gas inlet : This is where the gas supply is connected to the burner.
- Gas adjustment valve : is used to control the flow of gas
- Gas connector : is the part of the burner that connects to the gas source.
- Air holes : Air holes allow air to enter the burner to make a mixture of air and gas or other liquid fuel with air.
- The air regulator (Collar) : IIt is a short metallic cylindrical sleeve with two holes diametrically opposed to each other. Present around the ventilation holes, the main function of the collar is to control the amount of air entering the barrel.
- barrel or chimney : It is a metal tube about 5 inches long that rests on the base of the burner. It has two diametrically opposed holes near the bottom end which form the air vent. The barrel can be screwed to the base. The gas from the nozzle mixes with the air from the vent and burns at its upper end
- Flame stabilizer : This part helps to maintain a steady flame by providing a barrier to prevent the flame from flickering or extinguishing.
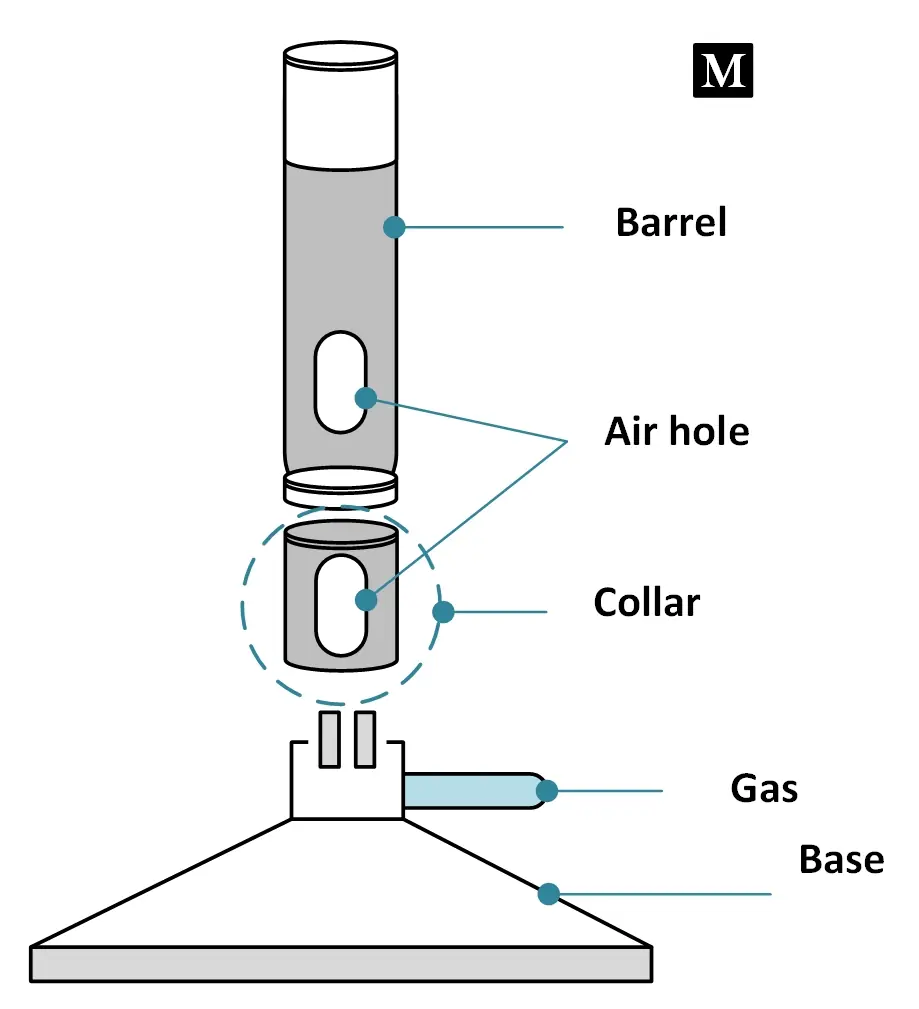
Bunsen burner parts
◉ Principle of Bunsen burner
Bunsen burners are normally fitted with a barbed fitting at the base of the chimney to allow a rubber tube to feed gas from a gas nozzle on the laboratory bench.
The Bunsen burner principle relies on its ability to mix gas (or other fuel) with oxygen before the mixture is ignited (creating a premix of air and gas before combustion). This is done by using an inlet valve at the bottom of the burner column which sucks in air by the Venturi effect, while the gas passes through a nozzle whose diameter is specific to the nature of the gas used. The mixture is then ignited at the top of the column.
The adjustable valve (collar) at the bottom dictates the amount of oxygen that enters the mix. With a closed valve, very little oxygen enters and a smoky yellow "low temperature" flame is produced. With the valve fully open, a hot, nearly colorless, roaring flame results.
◉ Fuel Sources
The choice of fuel source for a Bunsen burner depends on the specific application, availability of fuel sources, and safety considerations :
- Natural gas : it is a cost-effective and convenient option for locations with access to a natural gas supply. The gas line must be equipped with a shutoff valve that allows for safe and easy control of the gas supply.
- Propane gas : is commonly used as a fuel source for portable Bunsen burners, such as those used in field work or outdoor experiments.
- Butane gas : can be a convenient and portable fuel source for Bunsen burners in field work or outdoor settings where access to a natural gas supply may not be available.
Be sure to select the correct burner for the fuel source you will be using: it is dangerous to use a burner for one type of fuel with the other fuel
◉ Types of Bunsen Burners
The type of flame produced can be controlled by adjusting the air intake using the needle valve.
◉ There are several types of Bunsen burners to choose from depending on your gas source and experimental conditions :
- Standard version
- Micro Bunsen burners are miniature burners (smaller version of the previous one) and is used in smaller laboratory settings or for more delicate work.
- The Tirrill burner is a subtype of the Bunsen burner that features a wing nut at the bottom of the tube to regulate the gas supply. Minor gas adjustments can be made by turning this valve.
- The Meker burner is a subtype of the burner that is designed to produce an extremely hot and stable flame. The chimney is flared and the division of its flame by a grid at the top of the chimney gives a much shorter, more powerful flame (+ 1180°C) and quieter but with a higher gas consumption. It is commonly used in industrial settings.
- Fisher burner : This type is designed for use with natural gas. It is commonly used in analytical chemistry applications.
◉ What are the types of flame on a Bunsen burner ?
The type of flame produced can be controlled by adjusting the air intake using the needle valve. It is important to choose the appropriate flame for the specific laboratory application to ensure accurate and safe results.
◉ Safety Flame : The smallest and least hot flame, easy to see in a well-lit room, and helps others remember that your burner is on. Reaches temperatures of approximately 300 degrees, not used to heat materials for experiment.
Explanation : When the vent is closed, the air needed for the combustion reaction comes only from the area near the top of the burner, incomplete combustion, resulting in a bright yellow flame resembling a candle.
◉ Yellow flame : Produced when there is not enough air mixing with the gas, causing incomplete combustion. This flame is not commonly used in laboratory work as it can contaminate samples with soot.
◉ Medium blue flame : This particular flame on a burner can reach 500 degrees. Can be difficult to see in a bright room and is created when the air gap is partially open.
◉ Roaring blue flame : (It's the only flame that makes noise) This is the hottest flame, it can reach temperatures of 1400 degrees, with the hottest part of the flame just at the tip of the white cone in the middle of the flame.
Explanation : Increasing airflow to the burner produces more complete combustion and a hotter flame. The gas-air mixture is then ignited above the barrel. The result is a noisy, bluish-colored three-cone flame. This flame provides the highest possible burner temperature.

Types of flames